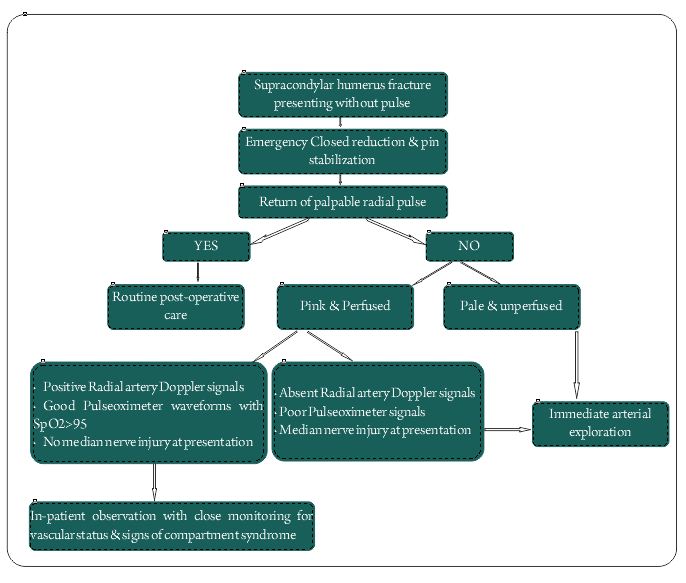
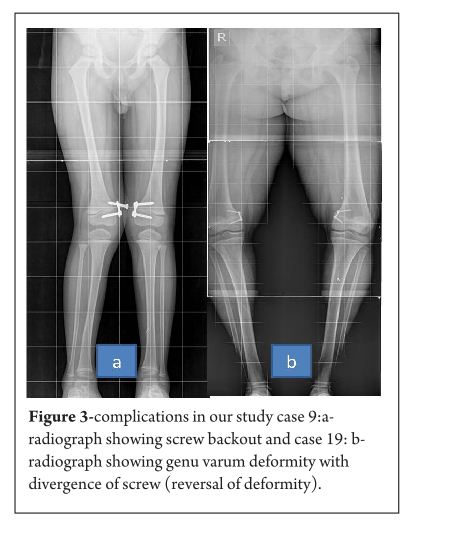
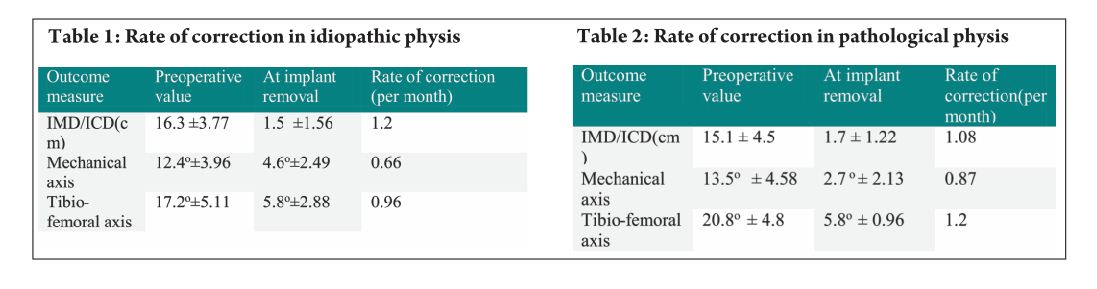
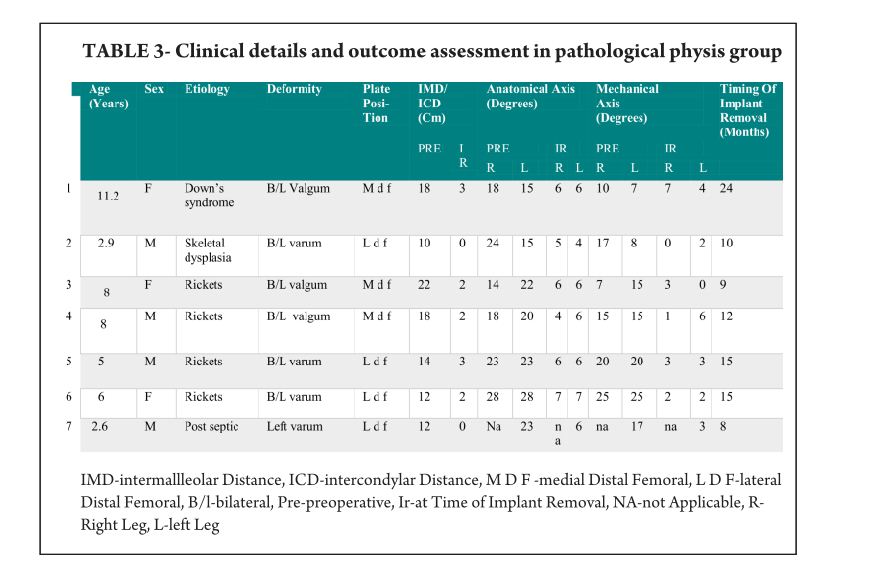
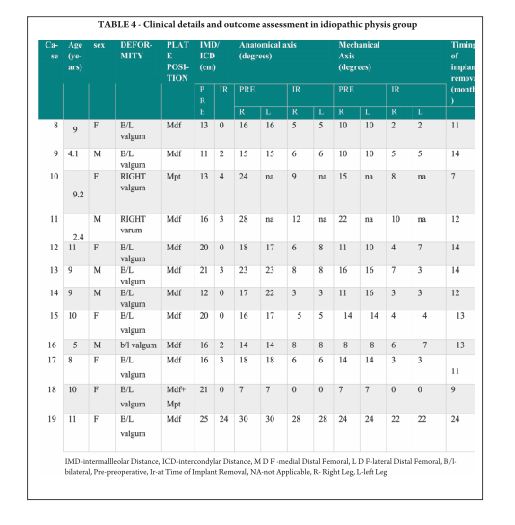
Pink Pulseless hand – Evaluation and Decision making: Is there a Consensus?
Vol 1 | Issue 1 | July-Sep 2015 | page:19-22 | Venkatadass K.
Authors : Venkatadass K[1].
[1] Consultant Paediatric Orthopaedic Surgeon, Ganga Medical Centre & Hospitals, Coimbatore, India.
Address of Correspondence
Dr. K. Venkatadass
Ganga Medical Centre & Hospitals, Coimbatore, India.
Email- venkatpedortho@gmail.com
Abstract
Background: The standard of care for the initial treatment of pulseless supracondylar fracture of the humerus is emergency closed reduction and percutaneous pin stabilization. Some of these patients remain pulseless even after closed reduction and pin fixation with a well perfused hand. The management of this so-called pink pulseless hand still remains controversial. The options described in the literature are either of the two extremes of just observation or exploration of the vessel and vascular repair if needed. There are no clear guidelines on when to explore a pink pulseless hand. This article reviews the current literature on this gray area with recommendations on the process of evaluation and decision-making in pink pulseless hand.
Keywords: Supracondylar humerus fracture, vascular injury, pink pulseless hand.
Introduction
The incidence of vascular injuries associated with displaced supracondylar fractures of humerus in children is about 10-20% [1,2,3]. It is reported to be more common in extension type fractures due to the close proximity of the proximal fragment to the neurovascular bundle [4,5]. In a child presenting with a pulseless supracondylar humerus fracture an urgent closed reduction with percutaneous pin stabilization is recommended. In majority of these patients the injured limb gets back the pulse and the hand appears well perfused after closed reduction and pin fixation. These patients are then treated as any other patient with supracondylar fracture humerus without any additional special precaution [3]. There is no confusion in the literature regarding the management of those patients in whom the limb remains pulseless, pale and unperfused after a closed reduction and stabilization [3]. Emergency exploration of the artery and arterial repair if needed to get back the circulation of the affected limb is the current recommendation. But there is still no consensus on the management of those limbs, which remain pulseless after closed reduction and pinning, but are pink and well perfused. The main reason for this confusion is lack of details of natural history of this entity [2]. The current literature on this enigmatous situation has been reviewed and a recommendation based on the available literature and our experience for the management of pink pulseless hand in supracondylar fracture humerus is presented.
Mechanism of vascular injury
The mechanism of injury to the neurovascular structures following supracondylar fracture of the humerus has been described in great detail by Meyerding as early as 1936 [6]. He was the first to study the configuration of the fracture in detail and propose that in extension type supracondylar fractures, the injuring force carries the distal fragment posteriorly stripping the posterior periosteum. The sharp anterior fragment pierces the anterior periosteum and brachialis and injures the neurovascular bundle, which lies in close proximity anteriorly. The vascular insult could either be due to compression from the fragment, spasm or thrombosis or rarely complete arterial transection. Most times the absence of pulse might just be due to the compression of grossly displaced fragments. A gentle closed reduction would relieve the compression and artery becomes pulsatile again. Louahem et al. [7 described 26 patients with a pink pulseless hand in a series of 210 patients with severely displaced supracondylar fractures. In 21 cases, the pulses returned immediately after closed reduction of the fracture.
Collateral circulation
The exuberant collateral circulation around the elbow has been credited with maintaining the vascularity of the limb in patients managed without vascular exploration [8]. The radial recurrent artery arises distal to the elbow and anastomoses with the radial collateral branch of the profunda brachii. The superior ulnar collateral artery is the other main descending collateral. It arises from the brachial artery, a little below the middle of the arm and anastomoses with the posterior ulnar recurrent and inferior ulnar collateral arteries [8].
What is pink pulseless hand?
There is variable use of this terminology in the literature. Some authors use this terminology to label those supracondylar fractures presenting without a pulse with a well perfused hand. While there is no controversy regarding the management of these fractures, more than 50% of these pink pulseless limbs would turn pulsatile just after closed reduction. The limb appearing pink and perfused on presentation mainly depends on the time duration since injury, as it needs some time for the collateral circulation to be established. None of the authors had looked into the time since injury and its correlation to perfusion status at presentation. Ideally, a pink pulseless limb is one that remains pink and well perfused without a palpable pulse following closed reduction and pinning of a pulseless supracondylar fracture [2]. These are the ones that pose management controversy as to whether it needs urgent exploration for the vessel or a closed monitoring for the vascular status and just observation. Though there is some evidence in the literature supporting immediate exploration, more recent evidence seems to be in favour of in-patient observation and close monitoring [3].
Role of Doppler
There has been increased interest in the role of colour Doppler in the process of evaluation and decision making for pink pulseless hands. White et al has recommended the use of colour Doppler to assess the severity of arterial injury following closed reduction and pinning to decide on further management. A colour Doppler evaluation of the brachial artery would help to differentiate between spasm, thrombosis and complete transection. It is important to remember the fact that there is no question of brachial artery injury in this scenario and the real issue is about the adequacy of the collateral circulation to maintain the viability and function of hand. Doppler evaluation of the brachial artery might infact increase the number of explorations of the artery. Valentini et al has reported the use of color-coded duplex scanning (CCDS) and ultrasound velocimetry (UV) of the hand as an additional tool for evaluation in all their patients with pink pulseless hands [9]. In their series, all seven patients with pink pulseless hand were found to have brachial artery injury by Doppler and all of them were treated by arterial repair. But, there are no clear guidelines on severity of arterial injuries on colour dopper and their management. Using Doppler to assess the radial artery in a pink pulseless hand helps to assess the adequacy of the collateral circulation. Weller et al [10] in their series of 54 pulseless supracondylar humerus fractures have documented that 26 patients regained the pulse after closed reduction, 20 remained pulseless after closed reduction but radial artery Doppler signals were picking up and 4 others had absent pulse as well as Doppler signals. All four were taken up immediate surgical exploration and found to have arterial injury requiring repair. All 20 patients who had pulse detected by Doppler but had no palpable radial pulse were observed. One of the 20 developed late ischaemia after nine hours and was taken up for surgical exploration. Shah et al have included triphasic radial artery doppler signal in their algorithm for decision making in pink pulseless limbs and recommends immediate surgical exploration for patients who do not have triphasic radial artery doppler signals.
Proponents for Immediate Exploration
White et al [11] after a systematic analysis of pink pulseless supracondylar fractures have concluded that there is significant arterial injury in 70% of patients and thus vascular exploration may limit the chances of late complications in these patients. They have also stated that with reported patency rates of more than 90% it is worthwhile considering exploration and arterial repair in these patients. Korompilias et al. [7] reported on five patients with a pink pulseless hand and recommended vascular exploration for the restoration of brachial artery patency, even in the presence of a viable well-perfused hand after an attempt at closed reduction. Copley et al [13] in their series of 17 patients with pulseless supracondylar fractures had a return of pulse in 14 of them following closed reduction. All the three patients were taken up for exploration and 2 of the fourteen patients who developed loss of pulse over the next 24 hours were also explored. They recommend immediate exploration if pulse is absent after closed reduction as a measure towards prevention of late complications. Blakey et al. found that twenty three of twenty-six patients with a pink, pulseless hand following initial management had some evidence of ischemic contracture, and they advocated for urgent exploration when the pulse does not immediately return after closed reduction [14]. Mangat et al [15] reported on the predictive value of co-existing median or anterior interiosseous nerve injury after studying a series of patients with nerve injury who underwent exploration. A significant relationship was found between preoperative median and anterior interosseous nerve deficits and vascular entrapment and tethering of the nerve at the fracture site. The authors recommended early exploration for patients with a Gartland type-III supracondylar fracture when they have coexisting anterior interosseous or median nerve palsy as the benefits of exploration outweigh the disadvantages. In a recent study by Scannell et al [17], the authors have tried to correlate the presence of median or anterior interosseous nerve with patency of brachial artery at long term follow-up in 20 twenty patients. In their series, median nerve palsy had good prediction of brachial artery occlusion while anterior interosseous nerve palsy did not predict brachial artery occlusion.
Proponents for Observation
Many authors are in favour of observation and close monitoring of the vascular status for pink pulseless hands. In contrast to the general belief that the literature in vascular surgery would be more in favour of arterial exploration, the recent papers in vascular surgery are recommending observation in case of pink pulseless hands [3]. Choi et al [18] presented the largest series of 33 patients with pink pulseless hands and concluded that in patients presenting with well perfused hand, fracture reduction and pinning alone would be sufficient treatment. Scannell et al [17] have reported the long term results of their series of 20 patients of pink pulseless hand that were treated by observation. All 20 had good functional outcome except one who had chondrolysis of the distal humerus. They also recommended long term follow-up of these patients for radiographic evidence of osteonecrosis as three of their 20 patients with pink pulseless hands developed avascular necrosis of the trochlea. Weller et al [10] in their analysis of 20 patients with pink pulseless hands have concluded that lack of palpable radial pulse is not an absolute indication for arterial exploration if Doppler signals and capillary refill is good suggesting a well perfused hand. Matuszewski [19] has published his follow-up results of pulseless supracondylar humerus fractures have concluded that children who, after satisfactory closed reduction, have a well-perfused hand but absent radial pulse do not necessarily require routine exploration of the brachial artery. Sabharwal et al [20], in their follow-up study patients with pulseless supracondylar fracture who were treated with arterial exploration and revascularisation found a high rate of asymptomatic reocclusion and residual stenosis and hence opined that collateral circulation would have been adequate to maintain a viable extremity. Garbuz et al [21] in 1996 presented the outcome of treatment of supracondylar fractures with absent radial pulse from the Hospital for Sick Children, Toronto. In their series of 22 patients, five had pink perfused pulseless hands who were managed by close observation and all had excellent functional outcomes. They concluded that absent pulse is not an absolute indication for exploration, provided the hand remains well perfused and compartment syndrome does not develop.
Is there a Consensus?
Though the literature is filled with publications on pink pulseless hand in supracondylar fractures, there still seems to be no consensus on the management of this condition. The AAOS guidelines [22] for the management of supracondylar fractures of humerus in children published in 2010 stated that ”We cannot recommend for or against open exploration of the antecubital fossa in patients with absent wrist pulses but with a perfused hand after reduction of displaced pediatric supracondylar humerus fractures” as there was no strong evidence supporting either observation or exploration. Five years down the line, the question still remains unanswered as far as evidence goes. But, what has changed over the years is that now we have more objective ways of assessing the perfusion rather than just relying on pink colour of the hand and capillary refill. The use of Doppler ultrasound and pulseoximeter signals to assess the perfusion of limb have come into vogue [23]. The presence of associated median nerve injury is more predictive of a significant arterial injury and hence these patients should be considered for exploration.
Hence in the present scenario, three factors needs to be considered in the decision making process of pink pulseless hand:
1.Presence of radial artery Doppler signals
2.Presence of good pulseoximeter waveforms and oxygen saturation >95%.
3.Intact Median Nerve function.
If all the three criteria are met, the recommendation is to observe the child closely for circulation and symptoms of compartment syndrome. If all three are absent, it is an indication of poor perfusion and it is an indication for arterial exploration. The combination of absence of radial artery Doppler signals and absence of pulse oximeter signals again indicates poor perfusion and favors exploration. There is no evidence to comment on other scenarios of either isolated median nerve palsy or isolated absence of radial artery Doppler signals or pulse oximeter signals and their combinations. There are no studies, which have documented all these factors for all their patients, and we are not sure whether a limb can have absent radial artery Doppler signals with good pulse oximeter waveforms. These would rather be hypothetical situations and if someone comes across such a situation in clinical practice, the best would be to individually assess the case and decide for exploration versus observation. However, considering the complications and the reported incidence of significant arterial injuries up to 70% in patients without a positive radial artery Doppler signal, it may be a safer option to consider exploration in these patients. The use of Doppler of the brachial artery to know the severity of the arterial injury and taking it as a sole factor for considering exploration is not justifiable, as the limb can still have good collateral circulation. Thus in conclusion both clinical and diagnostic methods have to be taken into account while making a balanced decision in terms on observation or surgical exploration of a pink pulseless hand [3,24].
References
1. Schoenecker PL, Delgado E, Rotman M, Sicard GA, Capelli AM. Pulseless arm in association with totally displaced supracondylar fracture. J Orthop Trauma 1996; 10:410–415.
2. Robb JE. The pink, pulseless hand after supracondylar fracture of the humerus in children. J Bone Joint Surg Br. 2009 Nov;91(11):1410-2.
3. Badkoobehi H, Choi PD, Bae DS, Skaggs DL. Management of the pulseless pediatric supracondylar humeral fracture. J Bone Joint Surg Am. 2015 Jun 3;97(11):937-43.
4. Korompilias AV, Lykissas MG, Mitsionis GI, Kontogeorgakos VA, Manoudis G, Beris AE (2009) Treatment of pink pulseless hand following supracondylar fractures of the humerus in children. Int Orthop 33(1):237–241.
5. Matuszewski Ł. Evaluation and management of pulseless pink/pale hand syndrome coexisting with supracondylar fractures of the humerus in children. Eur J Orthop Surg Traumatol. 2014 Dec;24(8):1401-6.
6. MEYERDING HW. Volkmann’s ischemic contracture associated with supracondylar fracture of humerus. Journal of the American Medical Association, 1936:106:1139-1144.
7. Louahem DM, Nebunescu A, Canavese F, Dimeglio A. Neurovascular complications and severe displacement in supracondylar humerus fractures in children: defensive or offensive
strategy? J Pediatr Orthop B 2006; 15(1):51–57
8. Ramesh P, Avadhani A, Shetty AP, Dheenadhayalan J, Rajasekaran S. Management of acute ‘pink pulseless’ hand in pediatric supracondylar fractures of the humerus. J Pediatr Orthop B. 2011 May;20(3):124-8.
9. Benedetti Valentini M, Farsetti P, Martinelli O, Laurito A, Ippolito E. The value of ultrasonic diagnosis in the management of vascular complications of supracondylar fractures of the humerus in children. Bone Joint J. 2013 May;95-B(5):694-8.
10. Weller A, Garg S, Larson AN, Fletcher ND, Schiller JR, Kwon M, Copley LA, Browne R, Ho CA. Management of the pediatric pulseless supracondylar humeral fracture: is vascular exploration necessary? J Bone Joint Surg Am. 2013 Nov 6;95(21):1906-12.
11. White L, Mehlman CT, Crawford AH. Perfused, pulseless, and puzzling: a systematic review of vascular injuries in pediatric supracondylar humerus fractures and results of a POSNA questionnaire. J Pediatr Orthop. 2010 Jun;30(4):328-35.
12. Korompilias AV, Lykissas MG, Mitsionis GI, Kontogeorgakos VA, Manoudis G, Beris AE. Treatment of pink pulseless hand following supracondylar fractures of the humerus in children. Int Orthop. 2009 Feb;33(1):237-41.
13. Copley LA, Dormans JP, Davidson RS. Vascular injuries and their sequelae in pediatric supracondylar humeral fractures: toward a goal of prevention. J Pediatr Orthop. 1996 Jan-Feb;16(1):99-103.
14. Blakey CM, Biant LC, Birch R. Ischaemia and the pink, pulseless hand complicating supracondylar fractures of the humerus in childhood: long-term follow-up. J Bone Joint Surg Br. 2009 Nov;91(11):1487-92.
15. Copley LA, Dormans JP, Davidson RS. Vascular injuries and their sequelae in pediatric supracondylar humeral fractures: toward a goal of prevention. J Pediatr Orthop. 1996 Jan-Feb;16(1):99-103.
16. Luria S, Sucar A, Eylon S, Pinchas-Mizrachi R, Berlatzky Y, Anner H, Liebergall M, Porat S. Vascular complications of supracondylar humeral fractures in children. J Pediatr Orthop B. 2007 Mar;16(2):133-43
17. Scannell BP, Jackson JB 3rd, Bray C, Roush TS, Brighton BK, Frick SL. The perfused, pulseless supracondylar humeral fracture: intermediate-term follow-up of vascular status and function. J Bone Joint Surg Am. 2013 Nov 6;95(21):1913-9.
18. Choi PD, Melikian R, Skaggs DL. Risk factors for vascular repair and compartment syndrome in the pulseless supracondylar humerus fracture in children. J Pediatr Orthop. 2010 Jan-Feb;30(1):50-6.
19. Matuszewski Ł. Evaluation and management of pulseless pink/pale hand syndrome coexisting with supracondylar fractures of the humerus in children. Eur J Orthop Surg Traumatol. 2014 Dec;24(8):1401-6.
20. Sabharwal S, Tredwell SJ, Beauchamp RD, Mackenzie WG, Jakubec DM, Cairns R, LeBlanc JG. Management of pulseless pink hand in pediatric supracondylar fractures of humerus. J Pediatr Orthop. 1997 May-Jun;17(3):303-10.
21. Garbuz DS, Leitch K, Wright JG. The treatment of supracondylar fractures in children with an absent radial pulse. J Pediatr Orthop. 1996 Sep-Oct;16(5):594-6.
22. The treatment of pediatric supracondylar humerus fractures. AAOS Clinical Practice Guidelines Unit v1.0_092311. Summary of Recommendations. available from
http://www.aaos.org/research/guidelines/SupracondylarFracture/SupConFullGuideline.pdf
23. Soh RC, Tawng DK, Mahadev A. Pulse oximetry for the diagnosis and prediction for surgical exploration in the pulseless perfused hand as a result of supracondylar fractures of the distal humerus. Clin Orthop Surg. 2013 Mar;5(1):74-81.
24. Shah AS, Waters PM, Bae DS. Treatment of the “pink pulseless hand” in pediatric supracondylar humerus fractures. J Hand Surg Am. 2013 Jul;38(7):1399-403
.
| How to Cite this Article: Venkatadass K. Pink Pulseless hand – evaluation and decision making: Is there a consensus?. International Journal of Paediatric Orthopaedics July-Sep 2015;1(1):19-22. |