Vol 1 | Issue 1 | July-Sep 2015 | page: 33-37 | Sandeep Patwardhan, Kunal Shah, Ashok K Shyam, Parag Sancheti.
Authors : Sandeep Patwardhan[1], Kunal Shah[1], Ashok K Shyam[1], Parag Sancheti[1].
[1] Sancheti Institute for Orthopaedics and Rehabilitation 16, Shivajinagar, Pune, India.
Address of Correspondence
Dr. Kunal Shah
Sancheti Institute for orthopaedics and Rehabilitation
16, Shivajinagar, Pune, India.
Email-orthokunal@yahoo.com
Abstract
Background: Angular deformities around the knee joint in skeletally immature children are treated with methods of reversible hemiepiphysiodesis like staples, transphyseal screw and eight plate. Hemiepiphysiodesis using Eight plate has showed good results with advantage being faster correction, less complications and can be used in younger age.
Methods: The aim of this retrospective study is show the efficacy of eight plate application and its complication rate. Nineteen patients (37 physes) (unilateral: 3; bilateral: 16) with angular deformity were treated with eightplate application. Seven with pathological physes and twelve with idiopathic physes. Outcome assessment was done clinically with calculation of intermalleolar /intercondylar distance and radiologicaaly with mechanical and anatomical axis. Correction achieved was considered when anatomical/mechanical axis were within normal limits and intermalleolar/intercondylar distance was less than 5 cm.
Results: The average age of intervention was 7.4±2.96 years (range 2.4 -11.2years). Rate of correction of IMD/ICD was 1.14 cm per month. Rate of correction of mechanical axis was 0.76 o per month. Rate of correction of anatomical axis was 1.04o per month. The average duration of eight plate removal 12.4 months (range 7-24 months).There were two complications one patient with screw backout and other with overcorrection.
Conclusion: Reversible hemiepiphysiodesis using eight plate is and effective method with minimal complications and faster rates of correction. Idiopathic physes show faster rates of correction than pathological physes. Physeal growth arrest is not seen with eight plate application. Larger data and long term follow up is required to assess the rebound deformity after eight plate removal.
Keywords: Reversible, hemiepiphysiodesis, angular deformity, eight plate.
Introduction
Pathological angular deformities of knee are common childhood deformities. Majority of them are idiopathic while others are due to some local or systemic cause [1].They present with cosmetic deformity, mild discomfort, gait disturbance, joint instability and limitation of activities or symptoms of causative disease. More importantly they predispose to early arthritic changes in the knee joint and secondary changes in hip and ankle joint [2, 3].Therefore it is important to identify them early and treat accordingly. Treatment depends mainly on cause of disease, age of child and amount of deformity. Corrective osteotomies once considered gold standard[3], are no longer advised in skeletally immature child, unless acute correction is required[4] or deformity is severe (>30o)[1]. Distraction osteogenesis using external fixator was used for gradual growth arrest, but it had several disadvantages like poor compliance, pin tract infections and longer time required to achieve correction [1].
Epiphysiodesis has emerged as the treatment of choice for angular deformity correction in skeletally immature patient with mild to moderate deformity[1,5]. Historically many methods for permanent and temporary epiphysiodesis were described [1]. Permanent methods depended on accurate timing of intervention to prevent overcorrection or under correction[5]. But none of the current methods of determining bone age are reliable [6, 7].Therefore reversible methods of epiphysiodesis have become the mainstay of treatment. It involves mainly staples, transphyseal screws and recently eight-plate has been used. Hemiepiphysiodesis with staples pose problems like migration, breakage and bending of implant, physeal growth arrest and rebound deformity[8].Transphyseal screws have shown fewer complications with implant and rebound phenomenon is less as compared to staples[5].However its reversibility is doubted by many as it cross the Physis[9,10]. Use of eight plate has shown promising results with fewer complications, faster correction and reversible growth [11, 12] yet literature is still sparse on use of this device. The purpose of this prospective study is to show the efficacy of hemiepiphysiodesis using eight-plate in correction of angular deformities around knee.
Material and Method
This is a retrospective study of 19 patients (37physes, 16 bilateral and 3 unilateral) with symptomatic angular deformity treated with 8 plate application. Out of the 19 patients there were 9 boys and 10 girls. Cause of angular deformity was rickets in 4, Down’s syndrome in 1, post septic in 1, skeletal dysplasia in 1 and idiopathic in 12. Genu varum was seen in 5 patients (8 physes) and genu valgum was seen in 14patients (29 physes).17 patients had 8 plate application in distal femoral physis, 1 patient had in proximal tibia and 1 patient had in both femur and tibia. Plates were applied in both tibia and femur to achieve faster rate of correction [11]. Surgical treatment was given to patients who were symptomatic or asymptomatic patients with age more than 4 years, with intermalleolar/intercondylar distance (IMD/ICD) more than 10 cm and/or mechanical axis more than 3° (valgum/varum). Contraindications to surgery included limbs with physiologic deformity, physeal arrest and maturity. Physiologic deformities were defined as genu varum in less than 2 years and genu valgum quantified by tibio-femoral angle less than 8° or IMD of less than 8 cm in age less than 4 years[14]. Physeal bar was defined as bony connection across physis, potentially affecting physeal growth [15]. The upper limit of age for surgical correction was at least one year of growth remaining [7, 9] as assessed on carpal age. In patient nearing skeletal maturity, hand film was taken to quantify amount of growth remaining. Standing lower limb scanogram with patella facing anteriorly [16] was taken preoperatively and at final follow up to look for mechanical axis and anatomical axis. Radiographically, mechanical axis of lower limb was measured as angle between mechanical axis of femur (centre of femoral head to centre of knee joint) and mechanical axis of tibia (centre of knee joint to centre of ankle mortise).The centre of knee joint was used to determine the mechanical axis [9]. The normal mechanical axis was considered as 0±3°[13]. With radiolucency of the physis it was difficult to define the centre of the knee joint and in such cases the centre of the distal physis of femur and proximal physis of tibia were considered in measurement. Tibiofemoral angle was measured as angle between long axis of femur and long axis of tibia. Normal was considered as 6° [16].
Clinically, intermalleolar and intercondylar distance were measured with patient in standing position with both patella facing forwards and medial malleolus/medial condyles just touching each other both preoperatively and at final follow up. Preoperative data was collected from the patient’s hospital records and from the surgeons own database. All patients with rickets were treated with appropriate medical management. Correction was considered when mechanical axis/anatomical axis were corrected and IMD/ICD was less than 5cm.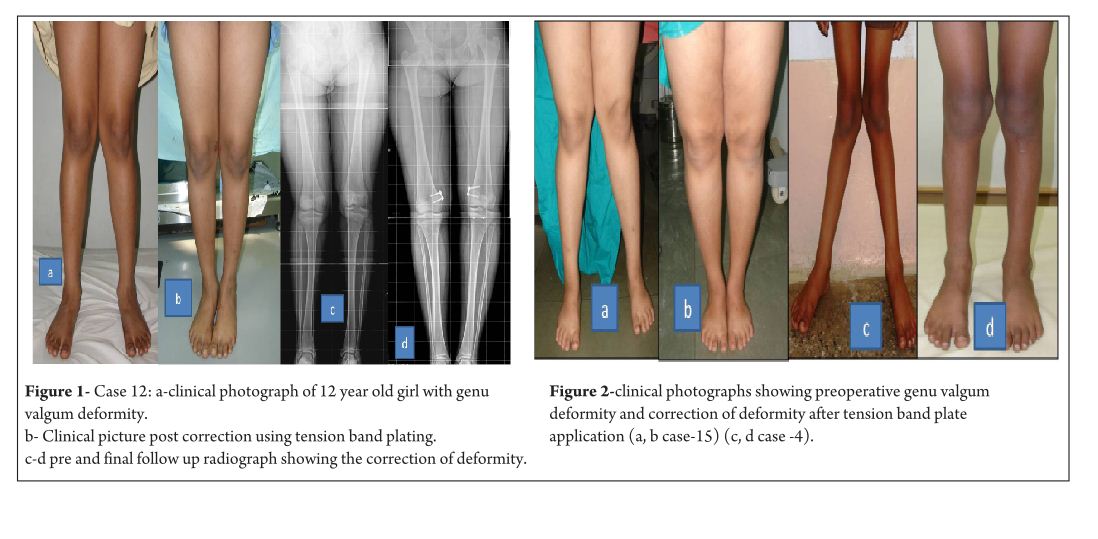
Surgical technique
With patient in supine position, tourniquet was applied to achieve haemostasis. Centre of Physis was marked using k wire under fluoroscopy guidance. Incision is taken and dissection done to reach the periosteum taking care that the periosteum is not breached and perichondrial blood supply is maintained. Plate inserted over the K wire and position confirmed under image intensifier, cannulated screws inserted parallel to Physis. Screw position checked under image intensifier in both antero-posterior and lateral view. Closure done and dressing applied. Postoperatively no immobilisation was required and patients were mobilised full weight bearing as tolerated from post op day 1. No walking support was required. Patients were followed up prospectively at every 3 month and knee radiographs were taken antero-posterior and lateral view to look for screw divergence and clinically IMD/ICD was measured.
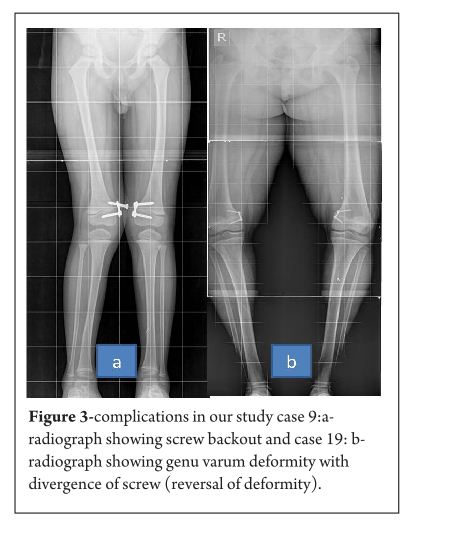
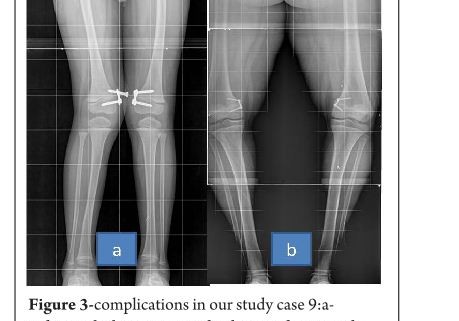
Results
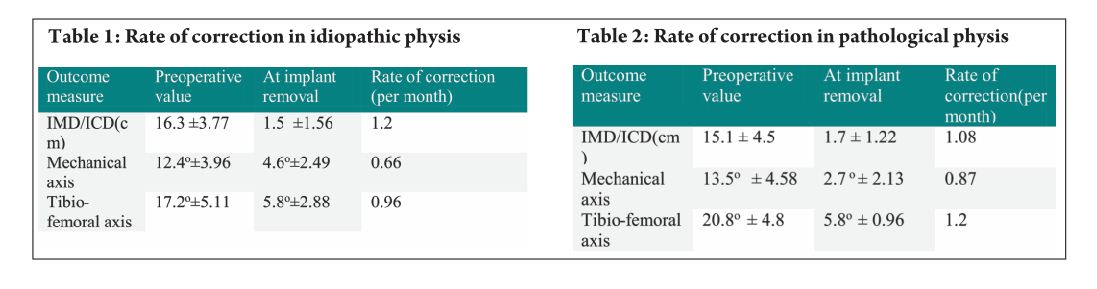
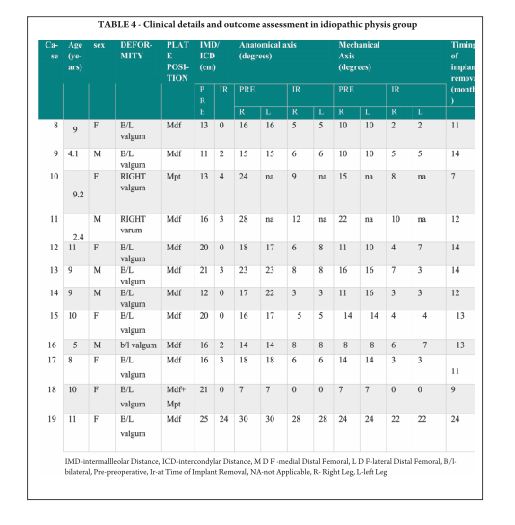
The average age of intervention was 7.4±2.96 years (range 2.4 -11.2years).The mean values of mechanical axis, tibiofemoral axis, IMD/ICD and duration of correction were calculated excluding the patient with overcorrection [case no 19] to avoid wide deviation from mean values. The mean preoperative IMD/ICD was 15.8cm ± 3.96 (range 10 cm to 22 cm) .The mean correction 1.6cm ± (range 0cm to 4 cm). Rate of correction was 1.14 cm per month. The mean preoperative mechanical axis was 13.4o± 5 (range7o to 25o).The mean post operative mechanical axis was 3.9o ±2.44 (range 0o to 10o). Rate of correction was 0.76 o per month .The mean preoperative tibiofemoral angle was 18.7o ±5.1 (range 7o -28o). The mean post operative tibiofemoral angle was 5.8o ± 2.29 (range 0o -12o). Rate of correction was 1.04o per month .Rate of correction in idiopathic and pathological physis group are depicted in table 1 and 2).The average duration of eight plate removal was 12.4 months (range 7-24 months). It was 13.3 months in pathological group and 11.8 months in idiopathic group. Figure 1 shows clinical and radiological correction in terms of IMD/ICD, mechanical axis and tibiofemoral axis. One patient was excluded from the data, a case of pathological genu valgum because the physis became fused before correction was achieved .The patient was treated with corrective osteotomy. Thus, emphasizing on preoperative planning in terms of age of intervention and aetiology deformity. There were two complications; one patient had screw back out which was revised. Other patient had reversal of deformity from 24° valgus to 22° varus due to delayed follow up [case 19]. She was treated with removal of 8 plate and is currently under observation for spontaneous correction. None of the patient had limb length discrepancy except (case 12) had 3 cm femoral shortening. Clinical data and outcome assessment in idiopathic and pathological group are depicted in table 1 and table 2.
Discussion
Physeal growth in child depends on variety of factors like biomechanical, hormonal and genetic [17]. Growth modulation using eight-plate depends on biomechanical growth modification based on Hueter-volkmann principle. Sustained compression parallel to physis leads to growth retardation and subsequent correction of deformity [9]. Eight-plate functions as flexible device which produces sustained compression at physis. The compression is not constant as the screws diverge with correction and with maximum divergence the plate bends, hence also called as tension band plate [11]. Eight-plate serves as non rigid implant with lateralisation of fulcrum for deformity correction. Thus, leading to faster rates of correction [ 2, 8]. Staples and transphyseal screws are rigid implants with centralised fulcrum for deformity correction [13]. They produce constant compression at physis. Thus they take longer time for deformity correction [2, 8]. Staples and transphyseal screws are rigid implants and if a prolonged duration is required for correction of deformity, they may cause physeal arrest [17]. In contrast eight plates are relatively flexible implants as it allows for screw separation. This is one of the reasons for decreased incidence of physeal arrest and makes it safer to use in younger children when compared to staples. In our study, three patients below the age of 3 years were treated successfully without any complications [case no 2, 7 and 11]. Clinical assessment of growth modulation by using IMD/ICD is reported for studies using staples [18] but not in studies using eight-plate. Since IMD/ICD was the major criteria to define the indication for surgery in our series, we have used the same as the primary outcome measure. We believe that IMD/ICD is an important clinical measure as majority of our patient were asymptomatic with cosmetic deformity and follow up parent counselling was easier. However standard values for particular age and race vary [14, 19].There can also be high rate of inter observer discrepancy and this is one of the limitations of the study. Radiographic measurements are used as outcome measures in papers on growth modulation using eight-plate like tibio-femoral axis, mechanical axis, joint orientation angles, mechanical axis deviation , articular –diaphyseal etc[2,10,11,20]We have used two radiological outcome measures, the hip knee ankle mechanical axis and the tibio-femoral angle. In our study we found that tibiofemoral angle and mechanical axis improved significantly. Standard antero-posterior and lateral radiographs of knee joint are useful in follow ups to see the effect of epiphysiodesis as seen with screw divergence. Joint orientation angles were not used, as scanograms become distorted due to magnification and parallax leading to false values[6].Also in young children it is difficult to visualise the distal femur and proximal tibia epiphyseal contours to accurately mark these angles. Ballal et al [11] showed that mean rate of correction of tibiofemoral axis 0.7°/month for distal femur and 0.5°/month in proximal tibia. Burghardt et al [20] showed mechanical axis correction of 1.73mm/month. In our study mean rate of correction of mechanical axis was 0.76°/month and tibio-femoral axis was 1.04°/month.
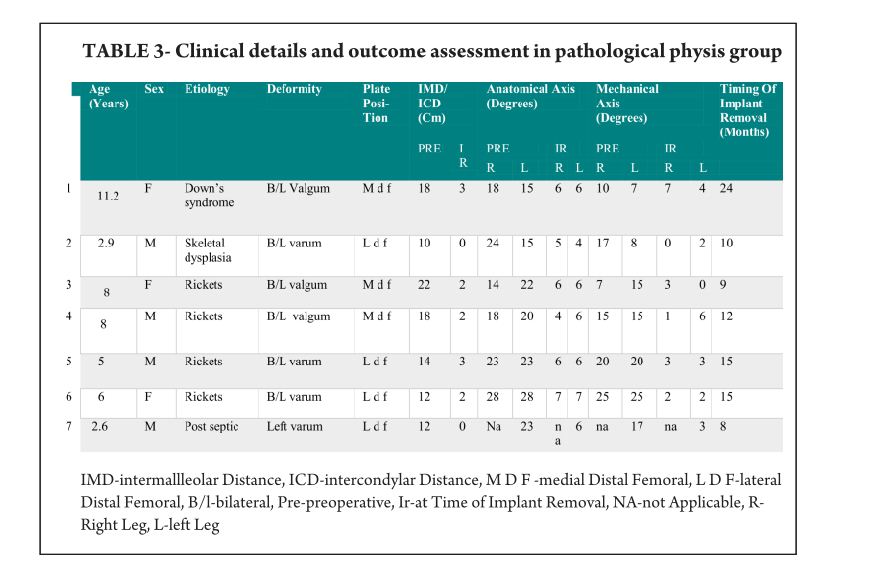
Rate of correction is faster in children less than 10 years as shown in study by Ballal et al [11]. In our study, due to smaller sample size of patients more than 10 years of age we could not assess this in our series. We did compared the rate of correction between the idiopathic and pathological group with former showing faster correction (Table 3 and 4). This corroborated with findings of Boero et al.No difference in rate of correction was encountered in terms of gender and type of deformity in our series. In our study two complications occurred, one patient had a screw back out (figure 3a) at 4 month after insertion, which was treated with revision of screw. However the rate of deformity correction was consistent with other patients in idiopathic physis group. We believe that the reason for back out may be placement of screw near the posterior cortex. Similar complications of screw loosening were seen in studies by Burghardt (n=1) [20] and Stevens (n=1) [2]. Other complication of overcorrection of deformity (figure 3b) was seen in one patient because of delayed follow up. This is justified as principally the sustained compression at physis will produce dynamic changes. Ballal et al [11] encountered one case of both screw and plate migration which was revised, such complication didn’t occur in our series. Several series encounter rebound growth after plate removal [2, 11, 20], we have not seen rebound of deformity in our patient with longest follow up of 2 years after plate removal. Complications related to implant like migration, breakage, bending of implants etc seen with staples are less common with eight -plate. Most dreaded complication of physeal arrest is not reported in our and other series [2, 3, 7, 10, 11, 20]. The threaded screws are less likely to extrude, especially in cartilaginous physis seen in younger age [8]. Screws are placed extra-periosteal and perichondrial blood supply is not hampered, so the chances of physeal arrest while insertion and removal are very less. Our study has drawbacks of small sample size and retrospective study design. However it does gives important inferences in terms of ability of 8 plates to correct the deformity and more importantly with minimal complications. However larger sample comparative studies will be required to establish the superiority of this method compared to other methods. Each author certifies that he or she has no commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
References
1. Celestre PC, Bowen RE. Correction of angular deformities in children- Current Orthopaedic Practice. 2009;20(6):641-647.
2. Stevens PM. Guided growth for angular correction: a preliminary series using a tension band plate. J Pediatr Orthop. 2007 Apr-May; 27(3):253-9.
3. Wiemann JM 4th, Tryon C, Szalay EA. Physeal stapling versus 8-plate hemiepiphysiodesis for guided correction of angular deformity about the knee. J Pediatr Orthop. 2009 Jul-Aug; 29(5):481-5.
4. Cho TJ, Choi IH, Chung CY, Yoo WJ, Park MS, Lee DY. Hemiepiphyseal stapling for angular deformity correction around the knee joint in children with multiple epiphyseal dysplasia. J Pediatr Orthop. 2009 Jan-Feb; 29(1):52-6.
5. Ghanem I, Karam JA, Widmann RF. Surgical epiphysiodesis indications and techniques: update. Curr Opin Pediatr. 2011 Feb; 23(1):53-9.
6. Friend L, Widmann RF. Advances in management of limb length discrepancy and lower limb deformity. Curr Opin Pediatr. 2008 Feb; 20(1):46-51.
7. Burghardt RD, Herzenberg JE, Standard SC, Paley D. Temporary hemiepiphyseal arrest using a screw and plate device to treat knee and ankle deformities in children: a preliminary report. J Child Orthop. 2008 Jun; 2(3):187-97.
8. Eastwood DM, Sanghrajka AP. Guided growth: recent advances in a deep-rooted concept. J Bone Joint Surg Br. 2011 Jan; 93(1):12-8.
9. Stevens PM. Guided growth of the lower extremities. Current Orthopaedic Practice. March/April 2011; 22(2):142–149
10. Schroerlucke S, Bertrand S, Clapp J, Bundy J, Gregg FO. Failure of Orthofix eight-Plate for the treatment of Blount disease. J Pediatr Orthop. 2009 Jan-Feb; 29(1):57-60.
11. Ballal MS, Bruce CE, Nayagam S. Correcting genu varum and genu valgum in children by guided growth: temporary hemiepiphysiodesis using tension band plates. J Bone Joint Surg Br. 2010 Feb; 92(2):273-6.
12. Stevens PM, Klatt JB. Guided growth for pathological physes: radiographic improvement during realignment. J Pediatr Orthop. 2008 Sep; 28(6):632-9.
13. DeBrauwer V, Moens P. Temporary hemiepiphysiodesis for idiopathic genuavalga in adolescents: percutaneous transphyseal screws (PETS) versus stapling. J Pediatr Orthop. 2008 Jul-Aug; 28(5):549-54.
14. Heath CH, Staheli LT. Normal limits of knee angle in white children–genu varum and genu valgum. J Pediatr Orthop. Mar-Apr 1993.
15.Paterson HA Epiphyseal growth plate fracture. Springer, Berlin 2007.
16. Paley D. Principles of Deformity Correction. Berlin, Germany: Springer; 2002.
17. Frost HM, Schönau E. On longitudinal bone growth, short stature, and related matters: insights about cartilage physiology from the Utah paradigm. J Pediatr Endocrinol Metab. 2001 May; 14(5):481-96.
18. Courvoisier A, Eid A, Merloz P. Epiphyseal stapling of the proximal tibia for idiopathic genu valgum. J Child Orthop. 2009 Jun; 3(3):217-21.
19. Omololu B, Tella A, Ogunlade SO, Adeyemo AA, Adebisi A, Alonge TO, Salawu SA, Akinpelu AO. Normal values of knee angle, intercondylar and intermalleolar distances in Nigerian children. West Afr J Med. 2003 Dec; 22(4):301-4.
20. Burghardt RD, Herzenberg JE. Temporary hemiepiphysiodesis with the eight-Plate for angular deformities: mid-term results. J Orthop Sci. 2010 Sep; 15(5):699-704.
21. Boero S, Michelis MB, Riganti S. Use of the eight-Plate for angular correction of knee deformities due to idiopathic and pathologic physis: initiating treatment according to etiology-J Child Orthop 2011;5(3):209-216.
| How to Cite this Article: Patwardhan S, Shah K, Shyam AK, Sancheti P. Growth Modulation in Children for Angular Deformity Correction around knee – Use of Eight Plate. International Journal of Paediatric Orthopaedics July-Sep 2015;1(1):33-37. |