Vol 1 | Issue 1 | July-Sep 2015 | page:3-5 | Sandeep V Vaidya.
Authors : Sandeep V Vaidya[1,2,3*].
[1] Children’s Orthopaedic Clinic, Thane.
[2] B J Wadia Children’s Hospital, Parel Mumbai
[3] Jupiter Hospital, Thane, Maharashtra, India
Address of Correspondence
Dr Sandeep V Vaidya
Director, Children’s Orthopaedic Clinic, Thane. India.
Email: drsvvaidya@gmail.com
Abstract
Diseases show a tendency to vary according to changing socio-economic trends and fractures too have shown this tendency. Paediatric supracondylar humerus fractures are one of the most common fractures seen by paediatric orthopaedic surgeons. There are few notable trends that have been reported and few other that I have personally noted in my practice and in practice of my colleagues. This article put together the changes reported in literature and tries to combine it with clinically relevant practical situations. Special focus is on fracture presentation and on decision making in management.
Keywords: Supracondylar Humerus fracture, classification, management.
Introduction:
Supracondylar humerus fractures in children are commonly seen in day to day practice. In this section, we study the epidemiology and changing trends of these fractures with respect to incidence, patient profile, types, modes of injury, treatment trends and complications.
Incidence:
Supracondylar humerus fractures (SHF) comprise 17% of all pediatric fractures and are second in frequency to forearm fractures. According to an epidemiological study, the incidence of fracture supracondylar humerus is 308/100000 per year in the general population. It is also the commonest pediatric fracture around the elbow. One epidemiological study identified supracondylar fractures in 206 out of 355 elbow fractures (58%) [1]. Barr reported a higher incidence of supracondylar humerus fractures during the vacations [2].
Age and sex:
If age distribution is considered, in the 0 to 7 year age group, SHF is easily the commonest fracture seen (28%) [3]. The mean age at which fracture supracondylar humerus occurs is 5 to 8 years [1,2]. Wilkins proposed that when a child falls on extended upper extremity, the patients who demonstrate hyperextension (cubitus recurvatum) of the elbow are more predisposed to have supracondylar fractures. The children who do not have hyperextension of the elbow tend to sustain fractures of the radius and the ulna, usually at the distal portion. Since ligamentous laxity with elbow recurvatum is seen in younger children, this explains the higher incidence of supracondylar humerus fractures in younger children and higher incidence of radius ulna fractures in older children.
Recently, there seems be increase in incidence of SHF in lower age group(less than 2 years). Fractures occurring in these very young children may pose a diagnostic dilemma because in many of these cases, the fracture line is extremely low and on plain radiographs may mimic a fracture lateral condyle humerus due to the largely cartilaginous component of the distal fragment. In such cases, additional imaging like MRI or arthrogram may be needed to differentiate these low supracondylar fractures from the lateral condyle fractures (Fig. 1). Another peculiarity of the low supracondylar humerus fractures is that such fractures can be complicated by Avascular necrosis of the trochlea with subsequent later sequelae.
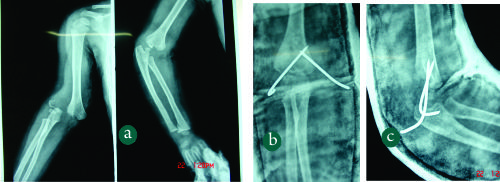
Figure 1: (Case Courtesy Dr Sandeep Patwardhan)1a: Elbow radiograph`of a 2 year old child with fall on outstretched hand. The fracture line is extremely distal and only a flake of metaphysis is seen.1b,c: The fracture was treated by closed reduction and K wire pinning .
In most of the earlier studies, the fracture occurred much more commonly in boys than in girls. However in most of the recent series, the frequencies in girls and boys seems to be equalizing. Some series have actually reported a higher incidence in girls than boys[1,2]. This changing sex distribution may be attributed to more active participation of girls in sports activities.
Mode of injury:
The cause of fracture supracondylar humerus is accidental fall while playing in most of the cases (60 to 80 %). Road traffic accidents account for 10 to 20% of SHF [2]. High velocity trauma can lead to fractures with metaphyseal comminution or in rare cases fractures with intercondylar extension.
Child abuse is an uncommon etiology of SHF[4]. However Strait and colleagues reported supracondylar fractures from abuse in three of 10 abused children under the age of 3, and cautioned that SHF should not be assumed to have non-abusive causes without careful consideration [5].
Types:
Extension type is the commonest type, flexion type is seen in 1 to 3% cases [6]. The patients in the flexion-type group (mean age, 7.5 years) are significantly older than those in the extension-type group (mean age, 5.8 years). The fractures in flexion-type group are also more probable to require open reduction (31%) than those in the extension-type group (10%). The flexion-type group had a significantly increased incidence rate of ulnar nerve symptoms (19% vs 3% in the extension-type group) and need for ulnar nerve decompression [7].
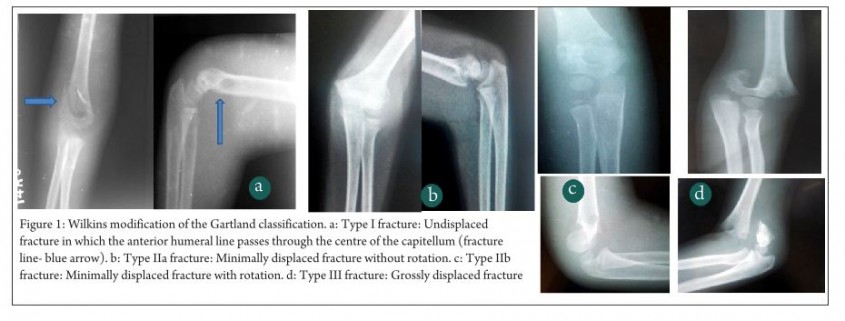
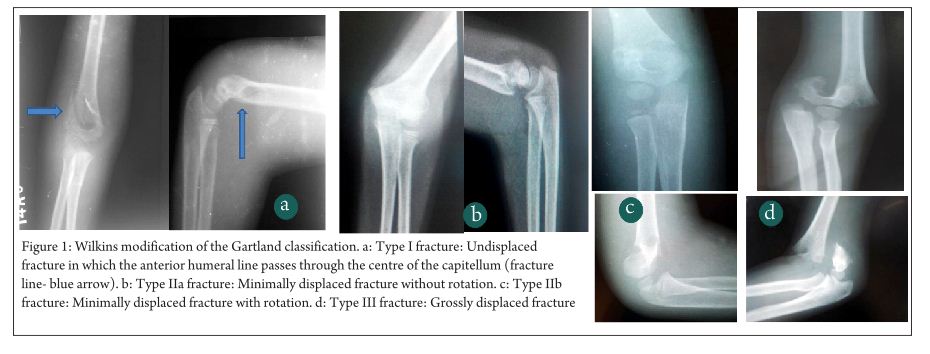
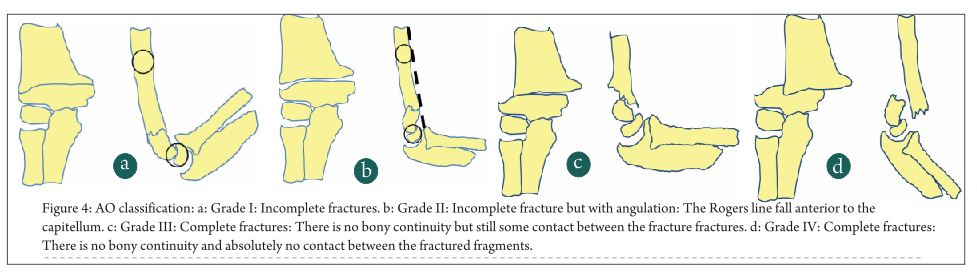
Gartland classification is the commonest classification system used to grade supracondylar humerus fracture. Grade 1 fractures are the commonest, followed by Grade 2 and then Grade 3 [1,2].
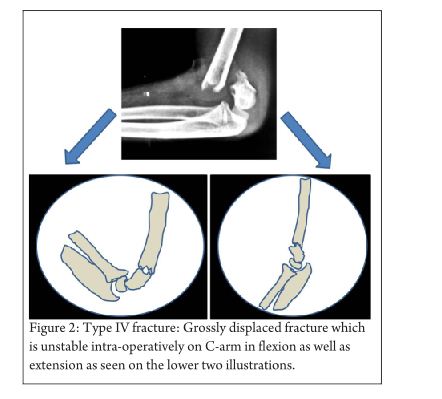
In addition to these 3 types, Leitch et al described a type 4 fracture with multidirectional instability (unstable in both flexion and extension). This fracture type was noted in 9 out of 297 displaced fractures. These fractures are associated with high velocity trauma, the periosteal sleeve is completely torn and special manoeuvres are needed for closed reduction- pinning [8].
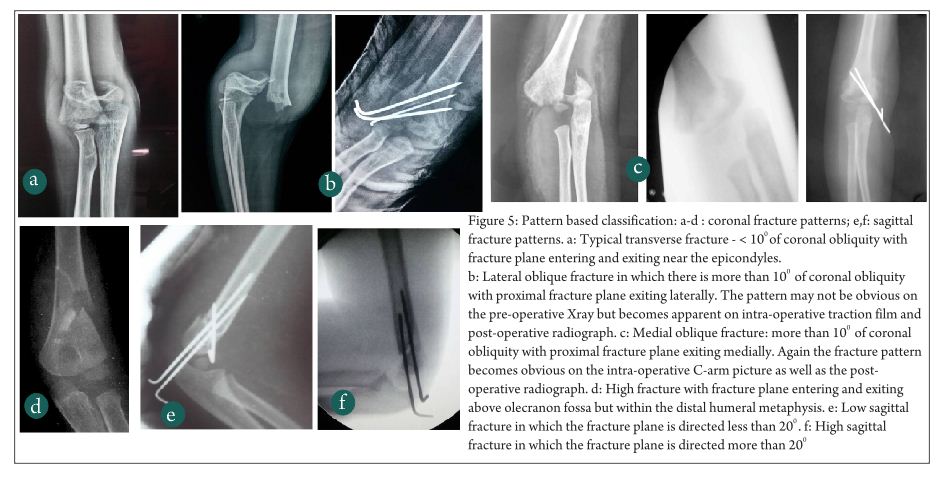
In extension type fractures the distal fragment may be displaced posteromedially or posterolaterally. Posteromedial displacement is commoner and seen in approximately 75% cases in most series. Posteromedial displacement of the distal fragment places the radial nerve at risk, whereas in fractures with posterolateral displacement the brachial artery and median nerve are at risk [9]. Bahk et al additionally classified extension type supracondylar fractures based on orientation of the fracture line in coronal as well as sagittal planes. In coronal plane, transverse fractures were the commonest (49%) followed by lateral oblique fractures (44%). Medial oblique (4%) and high transverse fractures (3%) were less common. Whereas transverse and lateral oblique fractures are amenable to lateral only pinning, the medial oblique and transverse fractures need to be fixed with medial-lateral cross pins [10].
High SHF are also being increasingly reported recently. Sen et al reported an incidence of high metaphyseal- diaphyseal supracondylar humerus fractures in 6 out of 182 fractures [11].
Treatment:
Blount in 1955 had cautioned against operative treatment in SHF citing the high incidence of complications following operative treatment [12]. However with significant advances in operative techniques and intraoperative imaging, operative treatment with Closed Reduction Percutaneous Pinning (CRPP) is easily the treatment of choice for displaced supracondylar humerus fractures [13]. Approximately 40% of SHF are treated operatively making it the commonest pediatric fracture to undergo operative treatment [2]. Cheng et al in an epidemiological study of 6493 fractures reported that the closed-reduction and percutaneous pinning rates for supracondylar humerus fractures increased 4.3 to 40% over a 10 year period from 1985 to 1995. The changes in treatment pattern were also accompanied by a corresponding decrease in the open-reduction rate and hospital stay periods from <10% to 38% of patients being discharged within 1 day of admission in the 10-year period [3].
The incidence of operative treatment is 0% in Grade 1 fractures, almost 50% for Grade 2 fractures, 100% for Grade 3 fractures and 100% for flexion type fractures. The incidence of open reduction is highest in flexion type fractures (50%) [2]. In an epidemiological study, out of 3235 children with displaced SHF treated operatively at a tertiary care children’s hospital at Toronto, 78.7% underwent operative treatment in the form of Closed Reduction Percutaneous Pinning (CRPP) whereas the remainder 21.8% underwent Open Reduction Internal Fixation (OR). There was a significant difference in the delay to surgery between the CRPP and OR groups [14]. In developed countries, there is a trend for more number of SHF are being treated by pediatric orthopaedic subspecialists. In New England, only 37% of SHF were treated by Pediatric Orthopaedic surgeons in 1991, this number rose to 68% in 1999. Kasser et al reported that in fractures treated by pediatric orthopaedic surgeons the length of hospitalization was lesser (1.4 ± 0.4 days) than for fractures treated by general orthopaedic surgeons (2.2 ± 0.6 days) [15]
Pin configurations, changing trends:
Pin configurations used by surgeons have shown a changing trend over the past decade. Several biomechanical studies published before 2005 revealed that crossed medial- lateral pin configurations are biomechanically stronger than lateral only pin configurations. Hence crossed medial- lateral pinning was preferred. However a major danger of the medial pin was iatrogenic ulnar nerve injury. Incidence of iatrogenic ulnar nerve injury with crossed medial- lateral pinning in various series has ranged from 0% to 6% [16,17]. Lyons et al reported iatrogenic ulnar nerve palsy in 19 out of 375 crossed medial- lateral pinning. 15 out of these 19 palsies recovered within 4 months after medial pin removal. However 4 palsies failed to recover, underwent ulnar nerve exploration and neurolysis [17]. A systematic pooled analysis of 32 trials comprising 2639 children suggests that there is an iatrogenic ulnar nerve injury for every 28 patients treated with the crossed pinning compared with the lateral pinning [16].
An inherent fallacy of the early biomechanical studies was that these studies were based on in-vitro findings wherein loads applied to create displacement were significantly higher than those which would be applied in-vivo wherein the fixation would be additionally supplemented with plaster slab application. Lee et al in their series of 61 consecutive lateral only pinning reported a zero incidence of loss of reduction as well as iatrogenic ulnar nerve palsy [18]. A randomized controlled study published in 2007 concluded that lateral entry only pinning did not result in increase incidence of loss of reduction as compared to crossed medial-lateral pinning [19]. A survey involving eight surgeons conducted in 2012 confirmed that this RCT had a significant influence on the surgeons’ practice. Five out of eight surgeons individually had a statistically significant change in their practice pattern for pin configuration. Except for certain selected fracture patterns, lateral only pinning is being increasingly used as the standard pin configuration for supracondylar humerus fractures [20].
Complications:
Complications of fracture supracondylar humerus include compartment syndrome, vascular injury, nerve injury (fracture related or iatrogenic) and malunion with cubitus varus deformity. The incidence of compartment syndrome is approximately 0.1% to 0.3% of all supracondylar humerus fractures [21]. Ipsilateral forearm fracture significantly increases risk of compartment syndrome [22]. In a study, the incidence of compartment syndrome was — % in fractures reduced and fixed within – hours of injury as compared to — % in fractures fixed after a delay of – hours.
The incidence of vascular injuries is approximately 20% and majority are associated with Grade 3 fractures [1, 23, 2]. Fractures with posterolateral displacement are more at risk for vascular injuries (approximately 65%) than fractures with posteromedial displacement (approximately 53%) [23]. If the hand is well perfused but pulseless, the great majority of the time fracture reduction is sufficient treatment. In contrast, patients presenting with a pulseless and poorly perfused hand have a nearly 50% chance of requiring vascular surgery and nearly 25% chance of developing a compartment syndrome [24, 25].
Nerve injuries are seen in approximately 4% fractures and majority are associated with Grade 3 fractures [1,2]. Overall, the most commonly injured nerve is median nerve (50%) followed by radial nerve (28%) followed by ulnar nerve (22%). The pattern of displacement is the most important risk factor in nerve injury. In fractures with median nerve palsy, posterolateral displacement is seen in 87% cases. In cases with radial nerve palsy, posteromdeial displacement Is seen in almost all cases [23] In flexion type, ulnar nerve is most commonly injured [7].
References
1. Houshian S, Mehdi B, Larsen MS. The epidemiology of elbow fracture in children: analysis of 355 fractures, with special reference to supracondylar humerus fractures. J Orthop Sci 2001;6(4):312-5
2. Barr LV. Pediatric supracondylar humeral fractures: epidemiology, mechanisms and incidence during school holidays. J Child Orthop. 2014; 8:167–170
3. Cheng, Jack CY, Ng, BKW, Ying, S. Y, Phil P. A 10-Year Study of the Changes in the Pattern and Treatment of 6,493 Fractures. 19(3), May/June 1999, pp 344-350
4. Kemp AM, Dunstan F, Harrison S, Morris S, Mann M, Rolfe K, Datta S, Thomas DP, Sibert JR, Maguire S. Patterns of skeletal fractures in child abuse: systematic review. BMJ 2008; 337:a1518
5. Strait RT, Siegel RM, Shapiro RA. Humeral fractures without obvious etiologies in children less than 3 years of age: when is it abuse? Pediatrics. 1995 Oct;96(4 Pt 1):667-71
6. Cheng JC, Lam TP, Maffulli N. Epidemiological features of supracondylar fractures of the humerus in Chinese children. J Pediatr Orthop B 2001;10(1):63-67
7. Mahan SD, May CD, Kocher MS. Operative Management of Displaced Flexion Supracondylar Humerus Fractures in Children. J Pediatr Orthop 2007;27:551-556
8. Leitch KK, Kay RM, Femino JD, Tolo VT, Storer SK, Skaggs DL. Treatment of multidirectionally unstable supracondylar humeral fractures in children. A modified Gartland type-IV fracture. J Bone Joint Surg Am. 2006 May;88(5):980-5.
9. Skaggs DL, Flynn JM. (2010) In Rockwood and Wilkins’ Fractures in Children. Philadelphia. Lippincott Williams and Wilkins. 514-515
10. Bahk MS, Srikumaran U, Ain MC, Erkula G, Leet AI, Sargent MC, Sponseller PD. Patterns of Pediatric Supracondylar Humerus Fractures. J Pediatr Orthop 2008;28:493-499.
11. Sen RK, Tripathy SK, Kumar A, Agarwal A, Aggarwal S, Dhatt S. Metaphyseo-diaphyseal junction fracture of distal humerus in children.
J Pediatr Orthop B 2012, 21:109–114
12. Blount WP. Fractures in Children. Baltimore: Williams and Wilkins, 1955
13. France J, Strong M. Deformity and function in supracondylar fractures of the humerus in children variously treated by closed reduction and splinting, traction and percutaneous pinning. J Pediatr Orthop. 1992:12(4): 494-498
14. Khoshbin A, Leroux T, Wasserstein D, Wolfstadt J, Law PW, Mahomed N, Wright JG. The epidemiology of paediatric supracondylar fracture fixation: A population-based study. Injury. 2014; 45: 701–708Khoshbin A, Leroux T, Wasserstein D, Wolfstadt J, Law PW, Mahomed N, Wright JG. The epidemiology of paediatric supracondylar fracture fixation: A population-based study. Injury. 2014; 45: 701–708
15. Kasser JR. Location of treatment of supracondylar fractures of the humerus in children. Clin Orthop Relat Res. 2005 May;(434):110-3
16. Slobogean BL, Jackman H, Tennant S, Slobogean GP, Mulpuri K. Iatrogenic ulnar nerve injury after the surgical treatment of displaced supracondylar fractures of the humerus: number needed to harm, a systematic review. J Pediatr Orthop 2010;30(5):430-6
17. Lyons, James P. M.D.; Ashley, Edwin M.D.; Hoffer, M. Mark M.D. Ulnar Nerve Palsies After Percutaneous Cross-Pinning of Supracondylar Fractures in Children’s Elbows. J Pediatr Orthop. 1998:18, 43-45
18. Lee YH, Lee SK, Kim BS, Chung MS, Baek GH, Gong HS, Lee JK. Three Lateral Divergent or Parallel Pin Fixations for the Treatment of Displaced Supracondylar Humerus Fractures in Children. J Pediatr Orthop 2008;28:417-422
19. Kocher MS1, Kasser JR, Waters PM, Bae D, Snyder BD, Hresko MT, Hedequist D, Karlin L, Kim YJ, Murray MM, Millis MB, Emans JB, Dichtel L, Matheney T, Lee BM. Lateral entry compared with medial and lateral entry pin fixation for completely displaced supracondylar humeral fractures in children. A randomized clinical trial. J Bone Joint Surg 2007;89(4):706-12
20. Mahan ST, Osborn E, Bae DS, Waters PM, Kasser JR, Kocher MS, Snyder BD, Hresko MT. Changing Practice Patterns: The Impact of a Randomized Clinical Trial on Surgeons Preference for Treatment
of Type 3 Supracondylar Humerus Fractures. J Pediatr Orthop 2012;32:340–345
21. Battaglia TC, Armstrong DG, Schwend RM. Factors affecting forearm compartment pressures in children with supracondylar fracture of the humerus. J Pediatr Orthop. 2002; 22(4): 431-439
22. Blackmore LC, Cooperman DR, Thompson GH. Compartment syndrome in ipsilateral humerus and forearm fractures in children. Clin Orthop and Relat Res. 2000; 376: 32-38
23. Campbell CC, Waters PM, Emams JB, Kasser JR, Millis MB. Neurovascular injury and displacement in type 3 supracondylar humerus fractures. J Pediatr Orthop. 1995;15(1):47-52
24. Choi PD, Melikian R. Skaggs DL. Risk Factors for vascular repair and compartment syndrome in the pulseless supracondylar humerus fracture in children. J Pediatr Orthop 2010;30:50-56
.
How to Cite this Article: Vaidya SV. Supracondylar Humerus Fractures in Children:
Epidemiology and Changing Trends of Presentation. International Journal of Paediatric Orthopaedics July-Sep 2015;1(1):3-5. |